9.9
1 Ratings
Thundercloud Consulting, LLC
Developer
14.9MB
Size
2021年03月16日
Update Date
Education
Category
4+
Age Rating
Age Rating
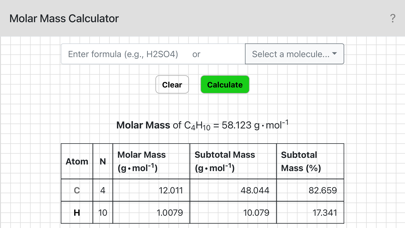
Chemistry - Molar Mass スクリーンショット
About Chemistry - Molar Mass
In chemistry the numbers of atoms, molecules, or formula units, amount of substance, in a sample is reported in the SI unit “mole” (abbreviated mol). One mole contains exactly 6.02214076 x 10^23 elementary entities.This number is known as Avogadro's constant.
A chemical substance may consist of atoms of a single element (e.g., O2), or of different elements (e.g., CO2). The molar mass of an element is the mass per mole of its atoms. The molar mass of a compound is the mass per mole of its molecules. The molar mass of an ionic compound is mass per mole of its formula units. To calculate the molar mass of a substance we add up the individual molar masses of the constituent atoms.
Note: In nature a sample of an element contains isotopes. Isotopes are atoms with the same number of protons in the nucleus (atomic number), but different masses. The difference in mass between isotopes is the result of different numbers of neutrons. The value of the molar mass of an element used in chemical calculations takes the natural abundance of isotopes into account (i.e., the molar mass is the weighted sum of the isotopic masses found in a typical sample).
To use this calculator enter a chemical formula and tap OK, or alternatively, select a sample molecule from the menu. For example, if you needed the molar mass of sulfuric acid you would enter H2SO4. Parentheses can also be used: the formula for pentane could be entered as CH3(CH2)3CH3 or CH3CH2CH2CH2CH3.
A chemical substance may consist of atoms of a single element (e.g., O2), or of different elements (e.g., CO2). The molar mass of an element is the mass per mole of its atoms. The molar mass of a compound is the mass per mole of its molecules. The molar mass of an ionic compound is mass per mole of its formula units. To calculate the molar mass of a substance we add up the individual molar masses of the constituent atoms.
Note: In nature a sample of an element contains isotopes. Isotopes are atoms with the same number of protons in the nucleus (atomic number), but different masses. The difference in mass between isotopes is the result of different numbers of neutrons. The value of the molar mass of an element used in chemical calculations takes the natural abundance of isotopes into account (i.e., the molar mass is the weighted sum of the isotopic masses found in a typical sample).
To use this calculator enter a chemical formula and tap OK, or alternatively, select a sample molecule from the menu. For example, if you needed the molar mass of sulfuric acid you would enter H2SO4. Parentheses can also be used: the formula for pentane could be entered as CH3(CH2)3CH3 or CH3CH2CH2CH2CH3.
Show More
最新バージョン 1.3 の更新情報
Last updated on 2021年03月16日
旧バージョン
Revised code and UX.
Show More
Version History
1.3
2021年03月16日
Revised code and UX.
1.2.1
2017年03月05日
Updated code base and user experience. New icon.
1.1.1
2015年10月02日
Added iOS 9 support.
1.1.0
2014年10月06日
iPod Touch / iPhone support. Bug fixes.
1.0.2
2013年05月06日
Code updates and interface makeover.
1.0
2012年03月07日
Chemistry - Molar Mass 価格
今日:
¥100
最低価格:
¥100
最高価格:
¥160
Chemistry - Molar Mass FAQ
ここをクリック!地理的に制限されているアプリのダウンロード方法をご参考ください。
次のリストをチェックして、Chemistry - Molar Massの最低システム要件をご確認ください。
iPhone
iOS 10.2以降が必要です。
iPad
iPadOS 10.2以降が必要です。
iPod touch
iOS 10.2以降が必要です。
Chemistry - Molar Massは次の言語がサポートされています。 英語