Chemical Equilibrium
$3.99
3.8for iPhone, iPad
Age Rating
Chemical Equilibrium 螢幕截圖
About Chemical Equilibrium
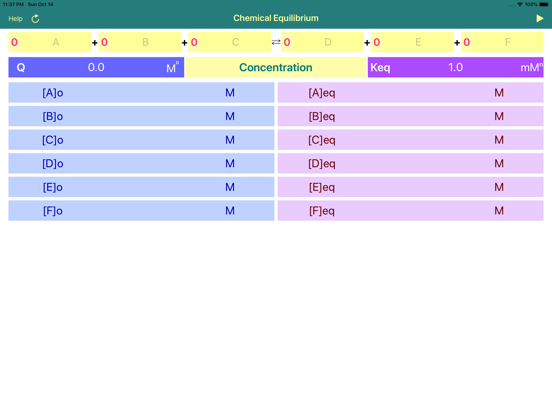
Chemical Equilibrium is a tool for accurate estimation of concentrations and pressures of the chemical reaction reactants and products at equilibrium. The application derives final, equilibrium parameters based on equilibrium constants (Kc and Kp), initial concentrations and stoichiometric coefficients. Additionally, app calculates reaction quotient for any given concentrations and stoichiometry.
For a general chemical reaction:
aA + bB = cC + dD,
the Reaction Quotient (Q) is defined by:
Q= (C^c * D^d) / (A^a * B^b)
where A, B, C, D – are compounds’ concentrations or pressures and a, b, c, d are stoichiometric coefficient*. Sum of powers determines the reaction order.
*Importantly, real system powers may differ from reaction coefficients, due to overall reaction mechanism complexity.
If Q is not equal to reaction equilibrium constant K (Kc for concentrations and Kp for pressures), then reaction is not at equilibrium and it will proceed (due to difference in forward and reverse reaction speeds) to the direction defined by Q.
If Q is less than K reaction will move towards products (C and D), in opposite case – reverse reaction will prevail.
Example:
For given reaction: 2A + 1B = 3D + 2E , initial reaction concentrations are Ao=0.1M, Bo=0.2M, Do=0.3M and Eo=0.4M and Kc=1 M^2.
the Reaction Quotient (Q) is defined by:
Q= (D^3 * E^2) / (A^2 * B)
Loading the data to “Chemical Equilibrium” app, Reaction Quotient is immediately calculated, giving Q= 2.16 M^2. Apparently Q>K, suggesting that reaction is too close to the products side and reverse reaction will prevail to bring the reaction back to the equilibrium.
The new equilibrium concentrations of reaction components can be derived from the equation:
Apparently, solving this equation is rather demanding task, that turns to be unnecessary, since “Chemical Equilibrium” app immediately returns the answer: Aeq= 118.5 mM, Beq= 209.2 mM, Deq= 272.3 mM and Eeq= 381.5 mM.
The application features:
•To start calculation user is required to fill in stoichiometric coefficients, initial concentrations (pressures) of available components, and equilibrium constant Kc (Kp).
•Reaction coefficients are actually representing powers and therefore define reaction order.
•If certain compound do not contribute to equilibrium calculations, like water in aqueous solution, then to exclude the compound it is required to long press on its name field. To signal about the change, the background colour of the non contributing field is altering. To revert compound to the normal state it is required to long press again the appropriate field. See the screenshot.
•To start calculation initially or after updating the concentration or coefficient fields, user is requested to tap Run button!
•Significant attention should be paid to concentration, pressure and equilibrium constant units. App automatically changes the set of available units as per order of reaction!
•The power of equilibrium constant units depicted as “x” is defined as (c+d-a-b), where a, b, c and d are the stoichiometric coefficients of the reaction.
•The basic conversions are as follows:
1 M= (1e3) mM
1 atm=101325 Pa
For a general chemical reaction:
aA + bB = cC + dD,
the Reaction Quotient (Q) is defined by:
Q= (C^c * D^d) / (A^a * B^b)
where A, B, C, D – are compounds’ concentrations or pressures and a, b, c, d are stoichiometric coefficient*. Sum of powers determines the reaction order.
*Importantly, real system powers may differ from reaction coefficients, due to overall reaction mechanism complexity.
If Q is not equal to reaction equilibrium constant K (Kc for concentrations and Kp for pressures), then reaction is not at equilibrium and it will proceed (due to difference in forward and reverse reaction speeds) to the direction defined by Q.
If Q is less than K reaction will move towards products (C and D), in opposite case – reverse reaction will prevail.
Example:
For given reaction: 2A + 1B = 3D + 2E , initial reaction concentrations are Ao=0.1M, Bo=0.2M, Do=0.3M and Eo=0.4M and Kc=1 M^2.
the Reaction Quotient (Q) is defined by:
Q= (D^3 * E^2) / (A^2 * B)
Loading the data to “Chemical Equilibrium” app, Reaction Quotient is immediately calculated, giving Q= 2.16 M^2. Apparently Q>K, suggesting that reaction is too close to the products side and reverse reaction will prevail to bring the reaction back to the equilibrium.
The new equilibrium concentrations of reaction components can be derived from the equation:
Apparently, solving this equation is rather demanding task, that turns to be unnecessary, since “Chemical Equilibrium” app immediately returns the answer: Aeq= 118.5 mM, Beq= 209.2 mM, Deq= 272.3 mM and Eeq= 381.5 mM.
The application features:
•To start calculation user is required to fill in stoichiometric coefficients, initial concentrations (pressures) of available components, and equilibrium constant Kc (Kp).
•Reaction coefficients are actually representing powers and therefore define reaction order.
•If certain compound do not contribute to equilibrium calculations, like water in aqueous solution, then to exclude the compound it is required to long press on its name field. To signal about the change, the background colour of the non contributing field is altering. To revert compound to the normal state it is required to long press again the appropriate field. See the screenshot.
•To start calculation initially or after updating the concentration or coefficient fields, user is requested to tap Run button!
•Significant attention should be paid to concentration, pressure and equilibrium constant units. App automatically changes the set of available units as per order of reaction!
•The power of equilibrium constant units depicted as “x” is defined as (c+d-a-b), where a, b, c and d are the stoichiometric coefficients of the reaction.
•The basic conversions are as follows:
1 M= (1e3) mM
1 atm=101325 Pa
Show More
最新版本3.8更新日誌
Last updated on 2023年12月04日
歷史版本
Compatibility update.
Show More
Version History
3.8
2023年12月04日
Compatibility update.
3.7
2022年01月20日
Compatibility update.
3.6
2021年06月15日
Compatibility update.
3.5
2021年01月22日
Compatibility update.
3.4
2020年07月24日
Compatibility update.
3.3
2020年05月31日
Compatibility update.
3.2
2020年03月30日
Compatibilty update.
3.0
2020年02月22日
Update to user interface.
2.9
2018年10月15日
Compatibility update.
2.7
2018年01月29日
Update adds possibility to exclude from calculations compounds that do not contribute to the reaction equilibrium, like water in aqueous solution. To exclude the compound it is required to long press on the name field. See the screenshot.
2.4
2018年01月26日
Compatibility update.
2.3
2017年11月27日
Compatibility update.
2.2
2017年10月22日
Compatibility update.
2.0
2017年02月09日
Number of components has been increased.
1.9
2016年07月11日
Compatibility update.
1.8
2016年05月09日
Compatibility update.
1.7
2016年03月08日
Compatibility update.
1.3
2015年10月12日
Compatibility update.
1.1
2015年08月18日
Compatibility update.
1.0
2015年04月04日
Chemical Equilibrium FAQ
Chemical Equilibrium在以下限制國家以外的其他地區可用:
Hungary,Kyrgyzstan,Macao,Tonga,Tanzania,Bulgaria,Cyprus,Czech Republic,Dominican Republic,Ecuador,Moldova,Trinidad and Tobago,Ukraine,Afghanistan,Cape Verde,Lebanon,Morocco,Mozambique,Turkmenistan,Uganda,British Virgin Islands,Cameroon,Mauritius,Turkey,Vanuatu,China,Kenya,Cambodia,Montserrat,Namibia,Nicaragua,Pakistan,Congo, Democratic Republic,Libya,Mauritania,Niger,Montenegro,Taiwan,Bolivia,Congo,Panama,Palau,Uzbekistan,Vietnam,Azerbaijan,Ghana,Gambia,Sri Lanka,Myanmar,Papua New Guinea,Paraguay,El Salvador,Grenada,Nigeria,Nauru,India,Mongolia,Slovenia,Kosovo,Israel,Kazakhstan,St. Lucia,Latvia,Cayman Islands,Angola,Croatia,Iraq,Mali,Turks and Caicos,Argentina,Dominica,Jordan,Maldives,Poland,Tajikistan,Tunisia,Burkina Faso,Costa Rica,St. Vincent & The Grenadines,Zimbabwe,Anguilla,Botswana,Suriname,Hong Kong,Malawi,Swaziland,Colombia,Lao Peoples Democratic Republic,Macedonia,Seychelles,Fiji,Micronesia,Yemen,Bermuda,St. Kitts and Nevis,Liberia,Oman,Rwanda,Uruguay,Zambia,Bhutan,Guyana,Honduras,Antigua And Barbuda,Albania,Guinea-bissau,Slovakia,Bahamas,Sao Tome & Principe,Barbados,Belarus,Jamaica,Nepal,Romania,Chad,Armenia,Bosnia & Herzegovina,Madagascar,Russia,Sierra Leone,Bahrain,Brazil,Estonia,Lithuania,Serbia,Venezuela,Benin,Belize,Senegal,Cote Divoire,Gabon,Malta,Algeria,Egypt,Georgia,Guatemala,Solomon Islands
Chemical Equilibrium支持English
點擊此處瞭解如何在受限國家或地區下載Chemical Equilibrium。
以下為Chemical Equilibrium的最低配置要求。
iPhone
Requires iOS 16.0 or later.
iPad
Requires iPadOS 16.0 or later.