Acid and Base Molarity Calc
Acid and Base Solution Tool
$2.99
1.3for iPhone, iPad and more
Age Rating
Acid and Base Molarity Calc Screenshots
About Acid and Base Molarity Calc
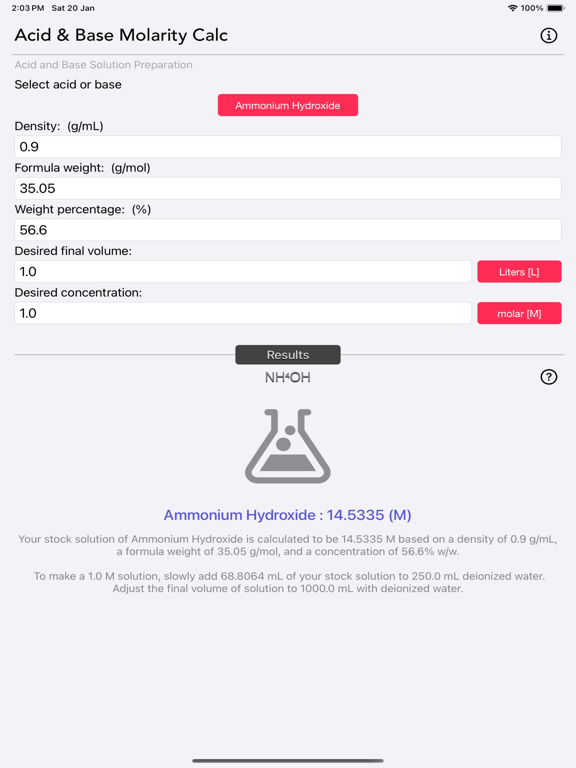
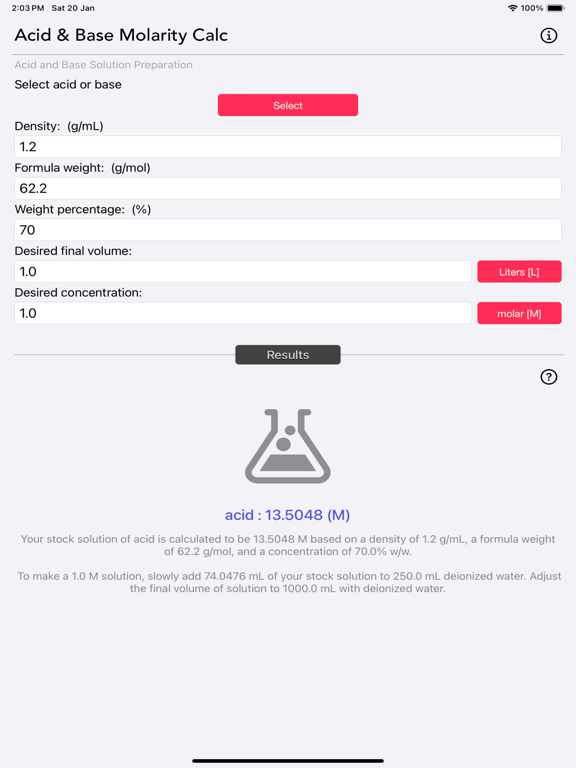
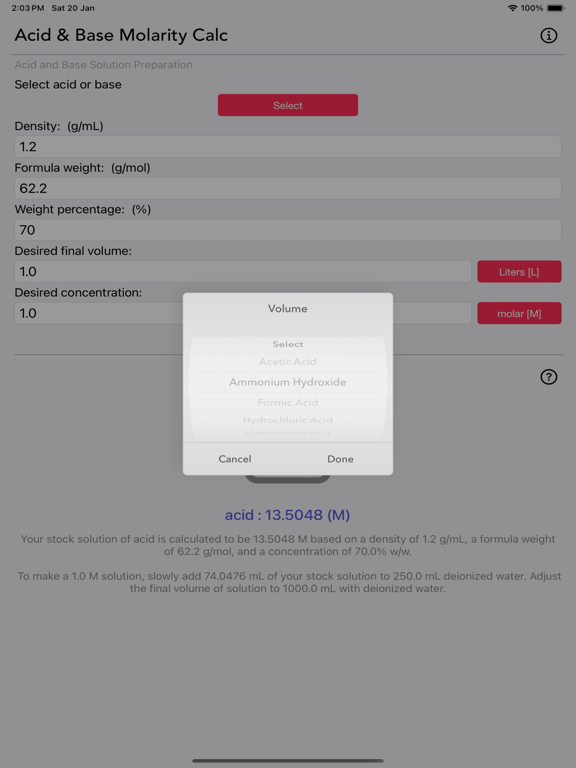
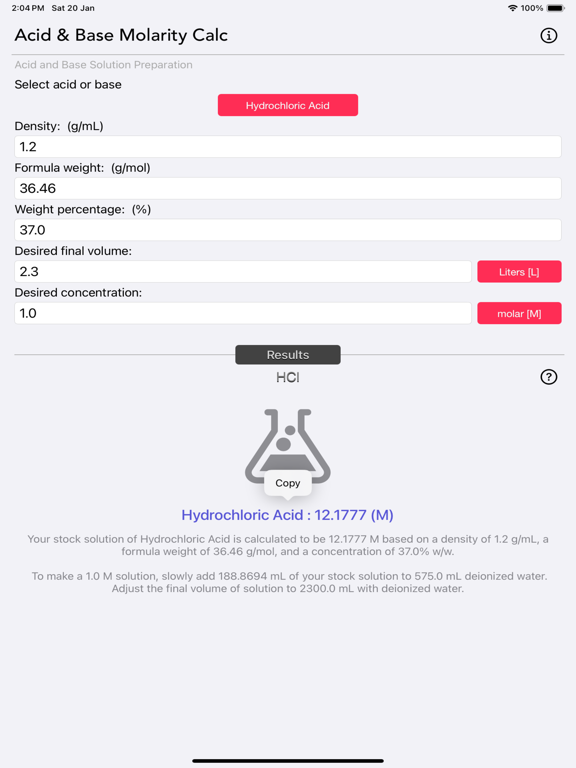
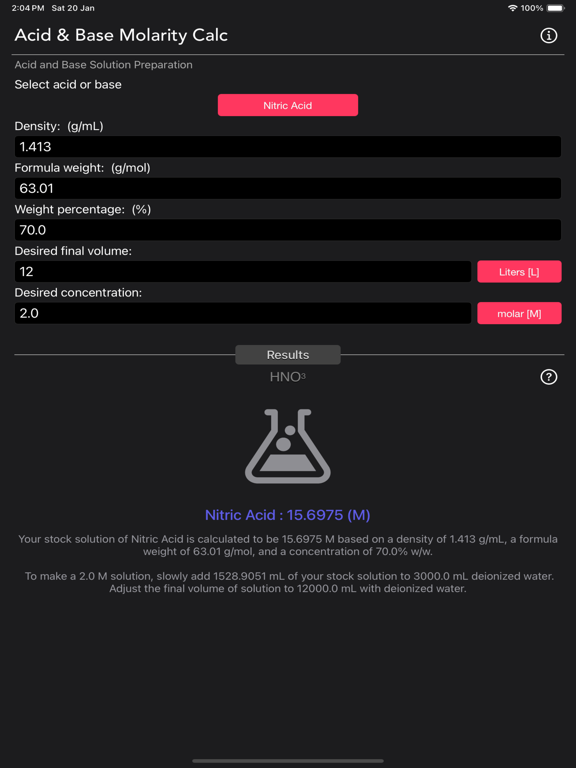
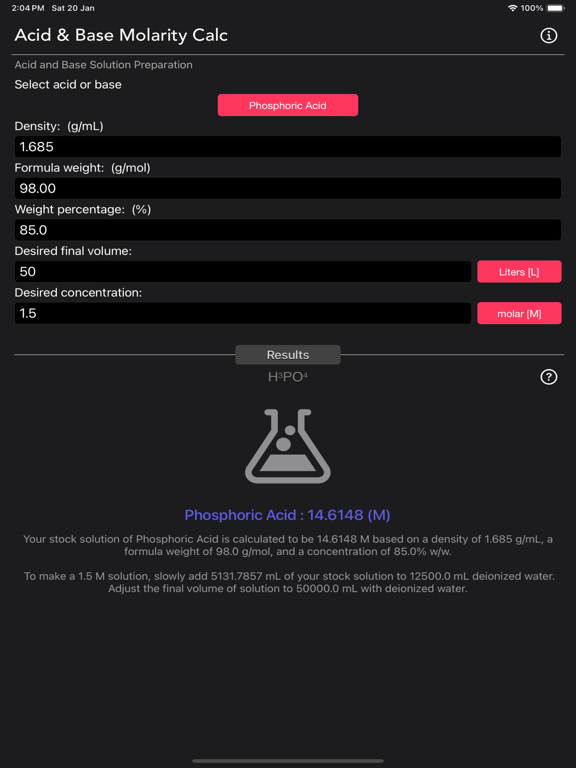
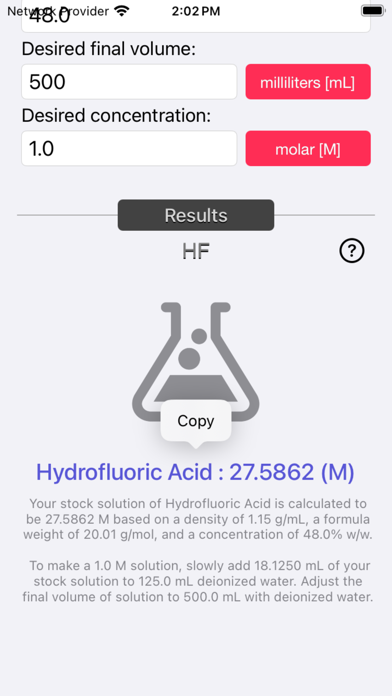
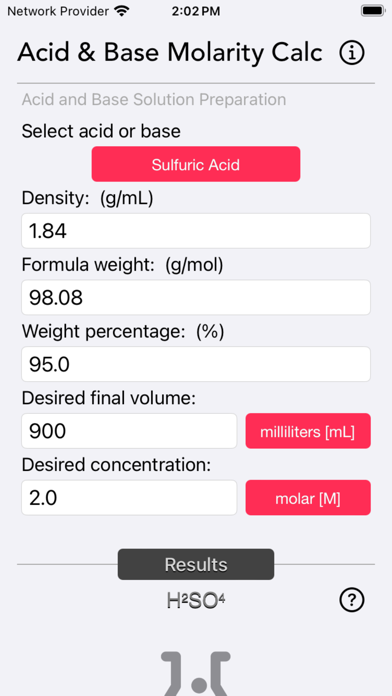
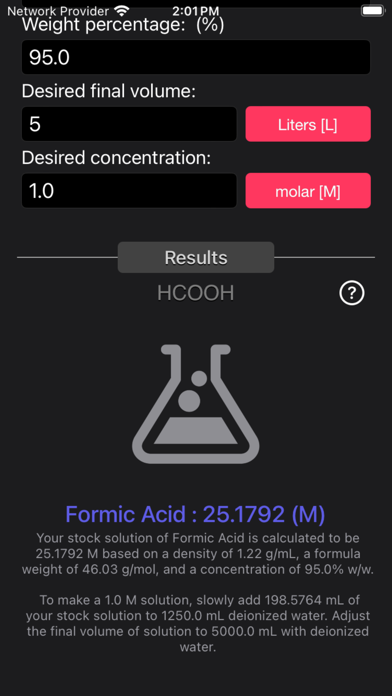
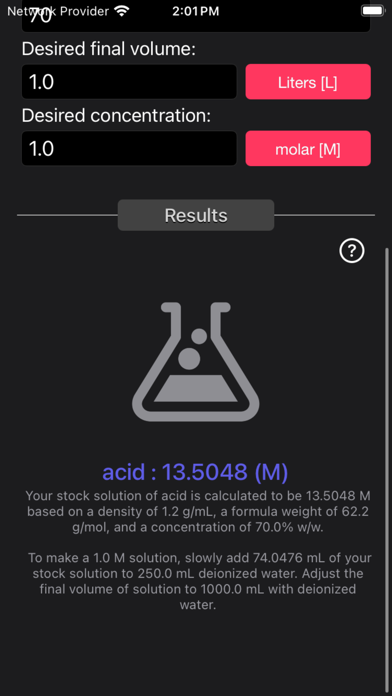
The Acid and Base Molarity Calculator provides lab-ready directions on how to prepare an acid or base solution of a specified Molarity (M) from a concentrated acid or base solution.
The molarity of a solution is defined as the number of moles of solute per liter of solution. Molarity facilitates easy dilution of a solution using the dilution formula, where the initial concentration multiplied by the initial volume equals the final concentration multiplied by the final volume (M1V1=M2V2). Multiplying the molarity by the volume gives the total moles of the material, which can then be diluted to any lower concentration.
Calculated results are copyable to other apps.
Formula:
Molarity= [(% x d) / MW] x 10
Where:
- % = Weight Percentage,
- d = Density,
- MW = Molecular Weight.
Molarity refers to:
1) The number of moles of solute per liter of solution.
2) The number of moles of a solute dissolved in a liter of solvent. Note: Be careful not to confuse molarity with molality. Molarity is represented by a small "m," whereas molality is represented by an uppercase "M."
3) The number of moles of solute in a liter of solution.
Example:
Calculate the Molarity for the given details.
Weight percentage = 250
Density = 50
Molecular weight/Formula Weight = 25
Solution:
Apply Formula:
Molarity = [(% x d) / MW] x 10
Molarity = 5000
Thanks for your support, and please visit nitrio.com for more apps for your iOS devices.
The molarity of a solution is defined as the number of moles of solute per liter of solution. Molarity facilitates easy dilution of a solution using the dilution formula, where the initial concentration multiplied by the initial volume equals the final concentration multiplied by the final volume (M1V1=M2V2). Multiplying the molarity by the volume gives the total moles of the material, which can then be diluted to any lower concentration.
Calculated results are copyable to other apps.
Formula:
Molarity= [(% x d) / MW] x 10
Where:
- % = Weight Percentage,
- d = Density,
- MW = Molecular Weight.
Molarity refers to:
1) The number of moles of solute per liter of solution.
2) The number of moles of a solute dissolved in a liter of solvent. Note: Be careful not to confuse molarity with molality. Molarity is represented by a small "m," whereas molality is represented by an uppercase "M."
3) The number of moles of solute in a liter of solution.
Example:
Calculate the Molarity for the given details.
Weight percentage = 250
Density = 50
Molecular weight/Formula Weight = 25
Solution:
Apply Formula:
Molarity = [(% x d) / MW] x 10
Molarity = 5000
Thanks for your support, and please visit nitrio.com for more apps for your iOS devices.
Show More
What's New in the Latest Version 1.3
Last updated on Feb 2, 2024
Old Versions
- Updated for the newest devices.
- Minor UI update.
- Minor bugs fixed.
- Minor UI update.
- Minor bugs fixed.
Show More
Version History
1.3
Feb 2, 2024
- Updated for the newest devices.
- Minor UI update.
- Minor bugs fixed.
- Minor UI update.
- Minor bugs fixed.
1.2
Dec 1, 2020
- Update for Newest Devices.
- Bug Fixed.
- Bug Fixed.
1.1
Apr 10, 2018
- Minor UI Update
1.0
Feb 25, 2018
Acid and Base Molarity Calc FAQ
Click here to learn how to download Acid and Base Molarity Calc in restricted country or region.
Check the following list to see the minimum requirements of Acid and Base Molarity Calc.
iPhone
Requires iOS 12.0 or later.
iPad
Requires iPadOS 12.0 or later.
iPod touch
Requires iOS 12.0 or later.
Acid and Base Molarity Calc supports English